Lupus Awareness Month
In this second October’s blog, we will be looking into Lupus in clinical trials: its symptoms, complications, treatments, incidence and focus on diversity.
Innovative Trials are passionate about ensuring our diverse population is adequately represented within medical research. Whether it is wanting to see more people from underrepresented communities choosing science as a career and pushing for greater patient diversity in clinical trials or focusing on what we are doing internally to celebrate and promote equality and diversity.
Throughout 2021, we have made a pledge to share our education and experiences with our clients and colleagues, to ensure inclusivity across the board. Each month we will be releasing communications in line with national and international awareness campaigns.
So far in our Awareness series, we have explored: Women’s Health; diversity in Cancer, Heart Disease and Scleroderma, Ovarian and Prostate Cancer, Autism Acceptance,Ramadan and Bowel Cancer, Stroke and Hepatitis, Mental Health, Pride month, Women with Alopecia, Psoriasis, Dementia, Disability and Black History Month

Introduction
Lupus Awareness Month takes place in the UK every October. This month is used to educate and raise awareness of lupus amongst the public as well as medical professionals, providing a deeper understanding of the symptoms and the impact the disease can have. [1]
What is Lupus
Lupus is an autoimmune disease as it causes the immune system to attack healthy cells in the body. It is mainly seen in females and can affect any part of the body. The disease often goes unnoticed for a long period of time as a result of the variety of symptoms that Lupus can produce, resulting in damage to various different organs such as the lungs, skin, heart, brain as well as the joints. Lupus is believed to be a genetic illness with no known cure, however symptoms can improve if treated early. [2]

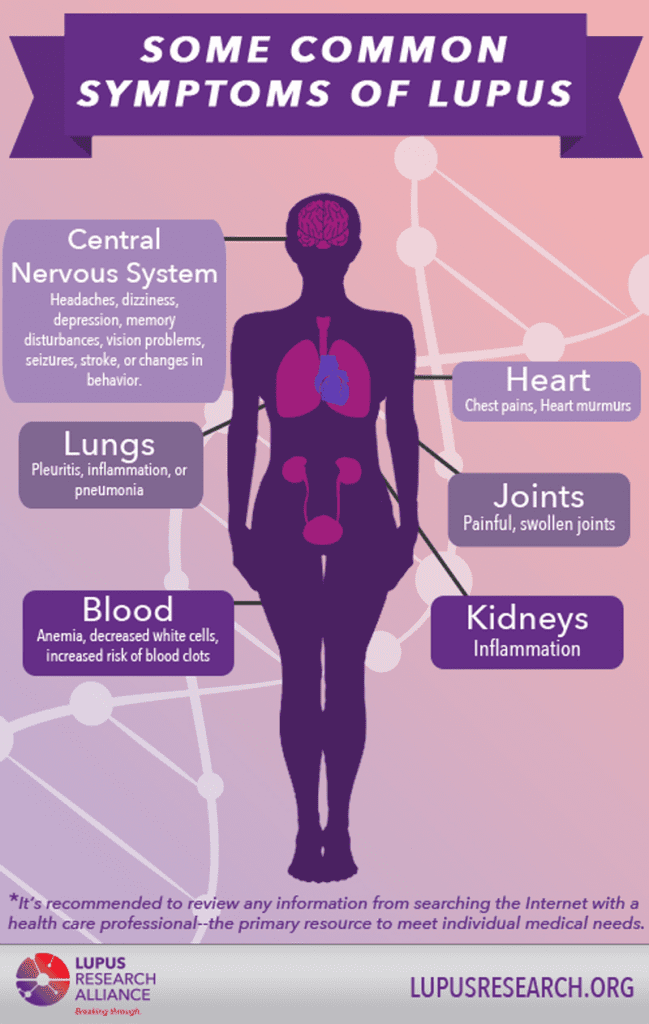
Lupus Symptoms
Lupus can often be difficult to diagnose as the signs and symptoms resemble those of other illnesses. The most distinctive sign of Lupus, however, is a facial rash that resembles butterfly wings across both cheeks. This occurs in many cases of lupus, but not all [3].
Cases of lupus differ from person to person. The symptoms will depend on the body systems that are affected by the disease, however the most common signs include:
- Fever
- Fatigue
- Joint pain, stiffness and swelling
- A butterfly-shaped rash on the face that covers the cheeks and bridge of the nose
- Rashes on other parts of the body
- Skin lesions that appear or worsen with sun exposure
- Shortness of breath
- Chest pain
- Dry eyes
- Headaches, memory loss and confusion
Complications
- Kidneys. Kidney failure has been found to be one of the leading causes of death among individuals suffering with lupus as it can cause severe kidney damage.
- Brain and central nervous system. Memory impairment is common among people with lupus as well as difficulties in expressing thought/communication. Dizziness, vision problems, a change in behaviour and even strokes or seizures can occur if the brain is affected.
- Blood and blood vessels. The illness may lead to blood complications such as a reduced number of healthy red blood cells (anemia) and an increased risk of bleeding or blood clotting. The blood vessels can also become inflamed.
- Lungs. Lupus also increases the chances of developing inflammation of the chest cavity lining, which can cause painful breathing. Bleeding into lungs and pneumonia also are possible.
- Heart. Inflammation of your heart muscle, your arteries or heart membrane can occur. This greatly increases the risk of cardiovascular disease and heart attacks [3].
Treatements
In general, lupus is treated with:
- Anti-inflammatory medicines such as ibuprofen
- Hydroxychloroquine for fatigue as well as skin and joint problems
- Steroid tablets, injections and creams for kidney inflammation and rashes
In severe cases, rituximab and belimumab can be used to treat lupus. These work on the immune system by reducing the number of antibodies in the blood. [4]
Incidence and Diversity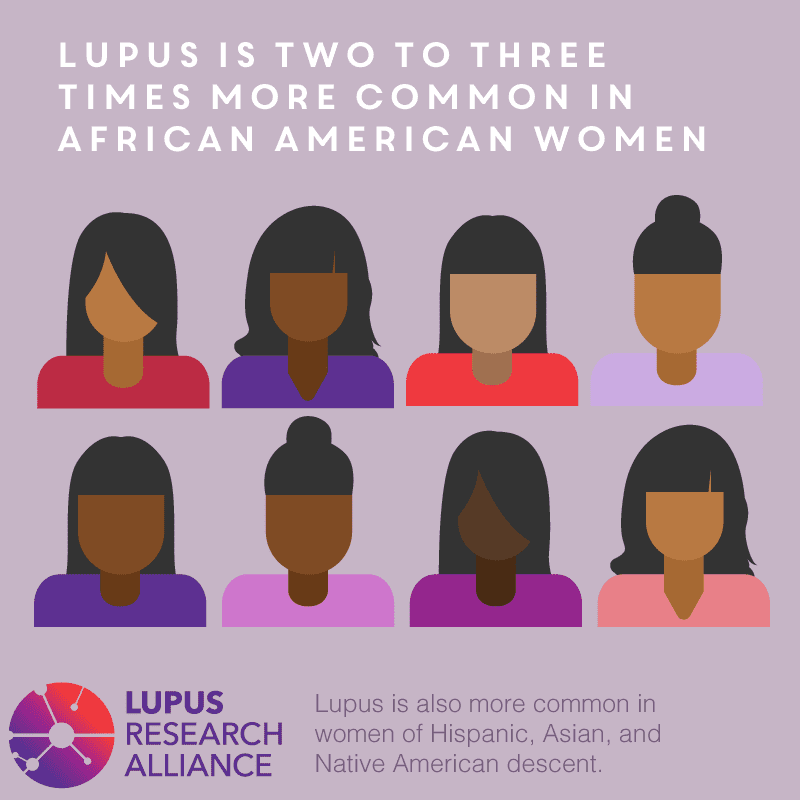
- The Lupus Foundation of America estimates that 1.5 million Americans, and at least five million people worldwide, have a form of lupus. [5]
- It is estimated based on available data on incidence that there are 16,000 new cases per year in the US alone. [5]
- It has been long established that 90% of individuals with lupus are women, with the illness being up to three times more prevalent in women of colour than Caucasian women. [5]
- Lupus has been previously ranked amongst the top 20 causes of death in all women, however this figure rose to distressing new levels as lupus is now recognized as the 5th and 6th leading cause of death among Black and Hispanic women aged 15-34. [6]
The lack of minority participation in clinical trials, including in lupus clinical trials, leads to a lack of data on the effectiveness, safety, and adverse effects of treatment within populations who have the highest incidence, prevalence, morbidity, and mortality from certain diseases.
Therefore, the participation from people from minority communities in clinical trials is essential to reduce lupus health disparities at both the individual and the systems levels. [7]
Innovative Trials Case Study
Innovative Trials has worked successfully on several Lupus studies. In one of the studies we have worked on, we found that sites that were supported by Innovative Trials screened on average 1.8x the number of patients screened at sites which did not receive Innovative Trials support. These supported sites also randomised on average 1.7x the number of patients randomised at sites which were not receiving our recruitment support.
References
- [1] Lupus UK (2021). Lupus Awareness Month. Retrieved September 2021 from https://www.lupusuk.org.uk/lupus-awareness-month/
- [2] Awareness Days (2021). Lupus Awareness Month 2021. Retrieved September 2021 from https://www.awarenessdays.com/awareness-days-calendar/lupus-awareness-month-2021/
- [3] Mayo Clinic. Lupus – Symptoms and causes. Retrieved September 2021 from https://www.mayoclinic.org/diseases-conditions/lupus/symptoms-causes/syc-20365789
- [4] NHS (2020). Treatments for Lupus. Retrieved September 2021 from https://www.nhs.uk/conditions/lupus/
- [5] Lupus Foundation Of America (2016) Lupus facts and statistics. Retrieved September 2021 from https://www.lupus.org/resources/lupus-facts-and-statistics
- [6] Lupus Research Alliance (May, 2021). Bolstering Diversity To Accelerate Research and Systemic Change. Retrieved September 2021 from https://www.lupusresearch.org/bolstering-diversity-to-accelerate-research-and-systemic-change/
- [7] Saira Sheikh, MD (2019). Diversity in Lupus Clinical Trials: Why Is It Important? Retrieved September 2021 from https://www.hmpgloballearningnetwork.com/site/rheum/diversity-lupus-clinical-trials-why-it-important?page=1
Equality, Diversity and Inclusion continues to be high on our agenda. We are working behind the scenes to push this forward. Keep your eyes peeled for more.
Find out how we’re working with clients to ensure greater patient diversity in clinical trials.
